Statistical Controversies in Reporting of Clinical Trials: Part 2 of a 4-Part Series on Statistics for Clinical Trials - ScienceDirect

Pediatric Clinical Research Networks: Role in Accelerating Development of Therapeutics in Children | Therapeutic Innovation & Regulatory Science
Post hoc analysis of SCORED trial sheds positive light on Inpefa for heart failure - Clinical Trials Arena

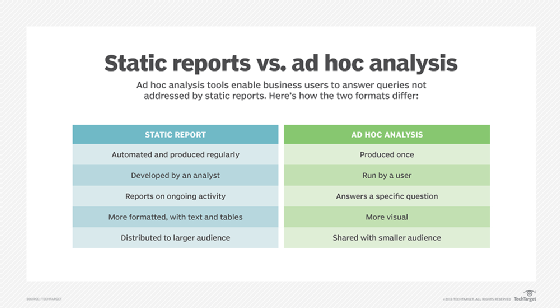
Conducting a clinical trial? 3 Kinds of statistical analysis you need to generate high-quality evidence | Editage Insights
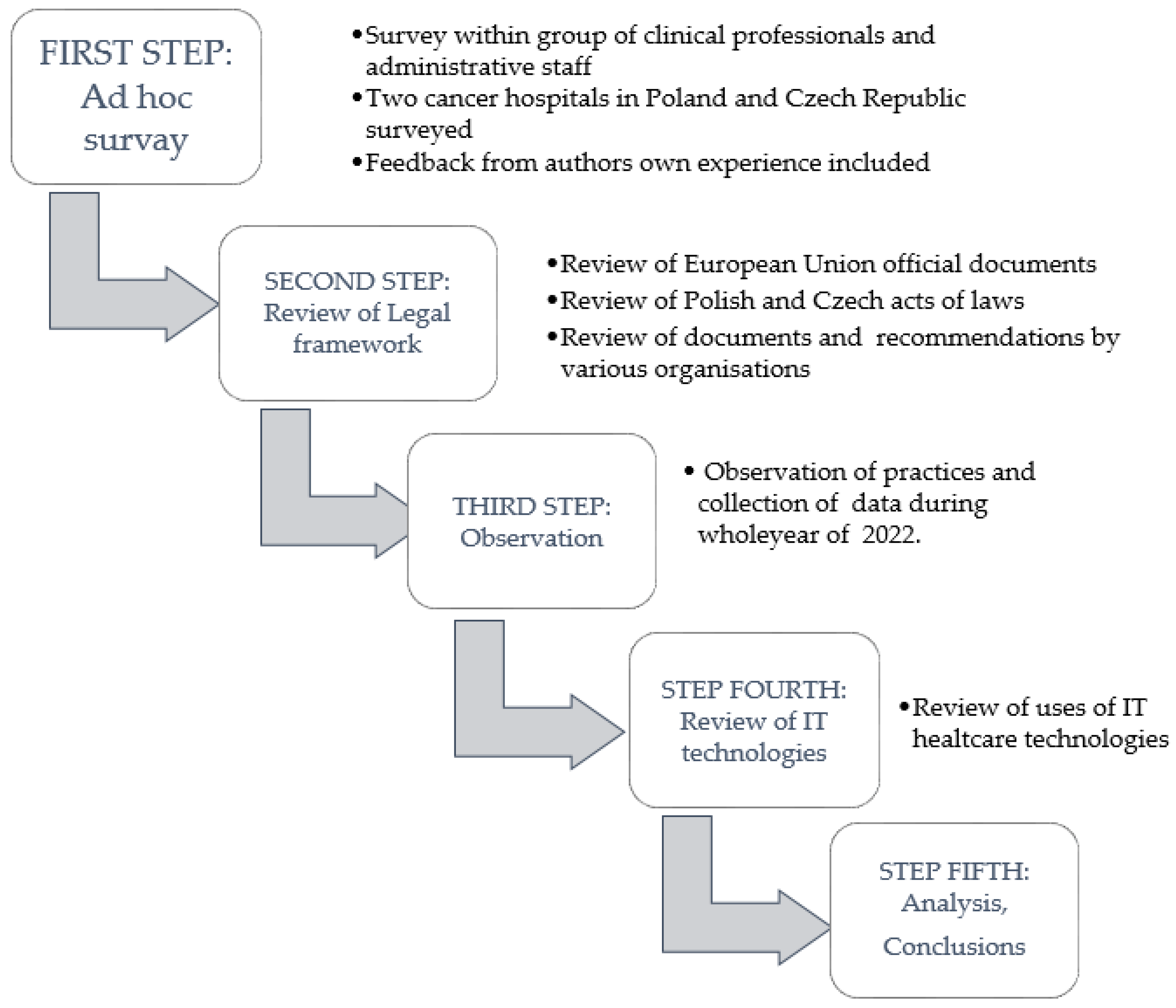
JPM | Free Full-Text | Management of Onsite and Remote Communication in Oncology Hospitals: Data Protection in an Era of Rapid Technological Advances
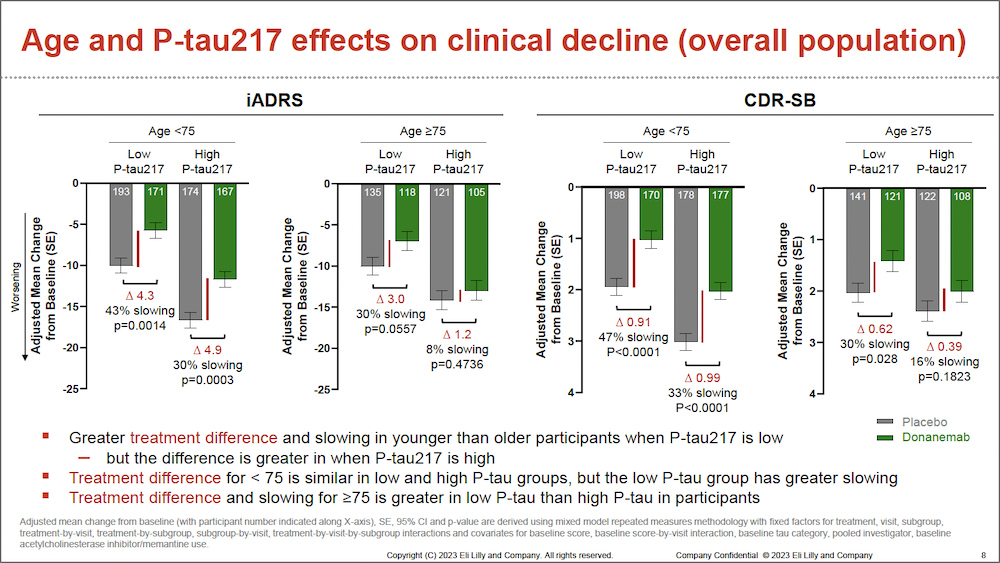
Donanemab Treatment Effects in Early Stages of Alzheimer Disease Detailed in New Post-Hoc Analysis from TRAILBLAZER-ALZ 2 - Practical Neurology

What I learnt from the Data Analytics for Pharma conference (and what "Ontology Driven" brings to the picture)

Neoadjuvant clinical trials provide a window of opportunity for cancer drug discovery | Nature Medicine
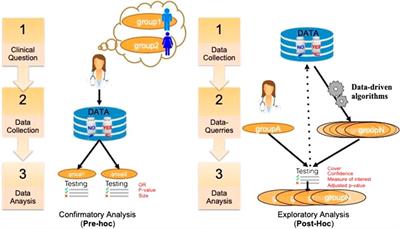
Frontiers | Q-Finder: An Algorithm for Credible Subgroup Discovery in Clinical Data Analysis — An Application to the International Diabetes Management Practice Study