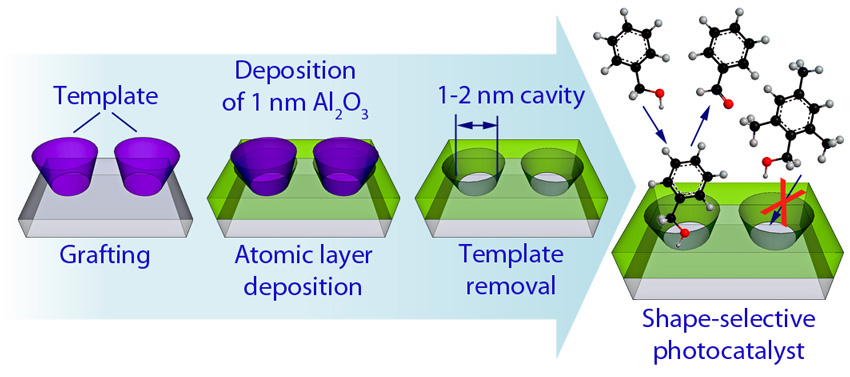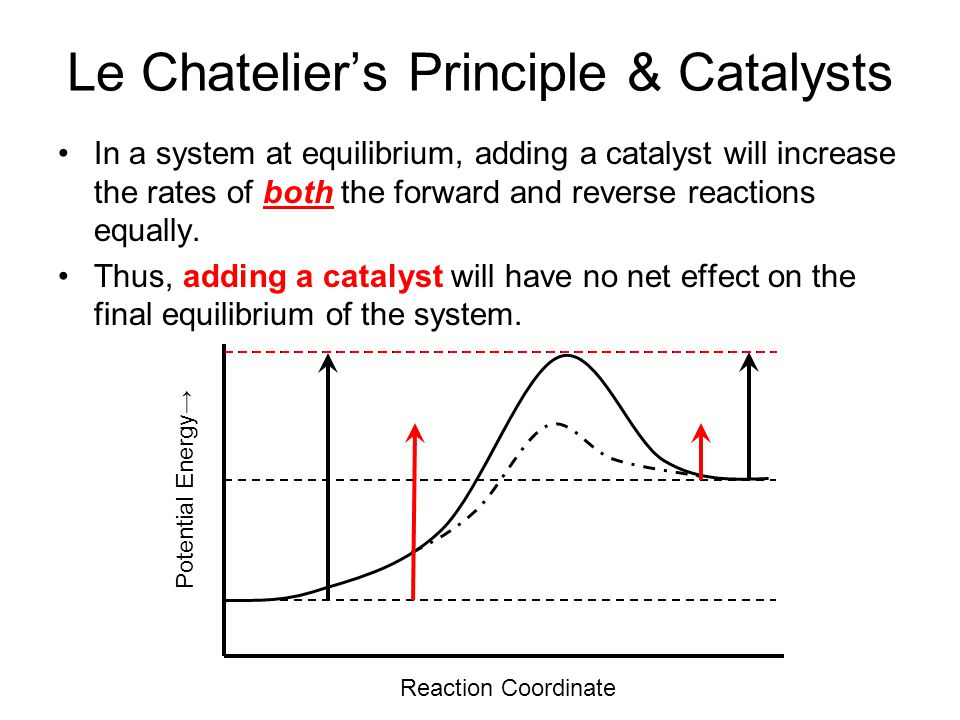
Le Chatelier's Principle & Catalysts. In a system at equilibrium, adding a catalyst will increase the rates of both the forward and reverse reactions. - ppt download
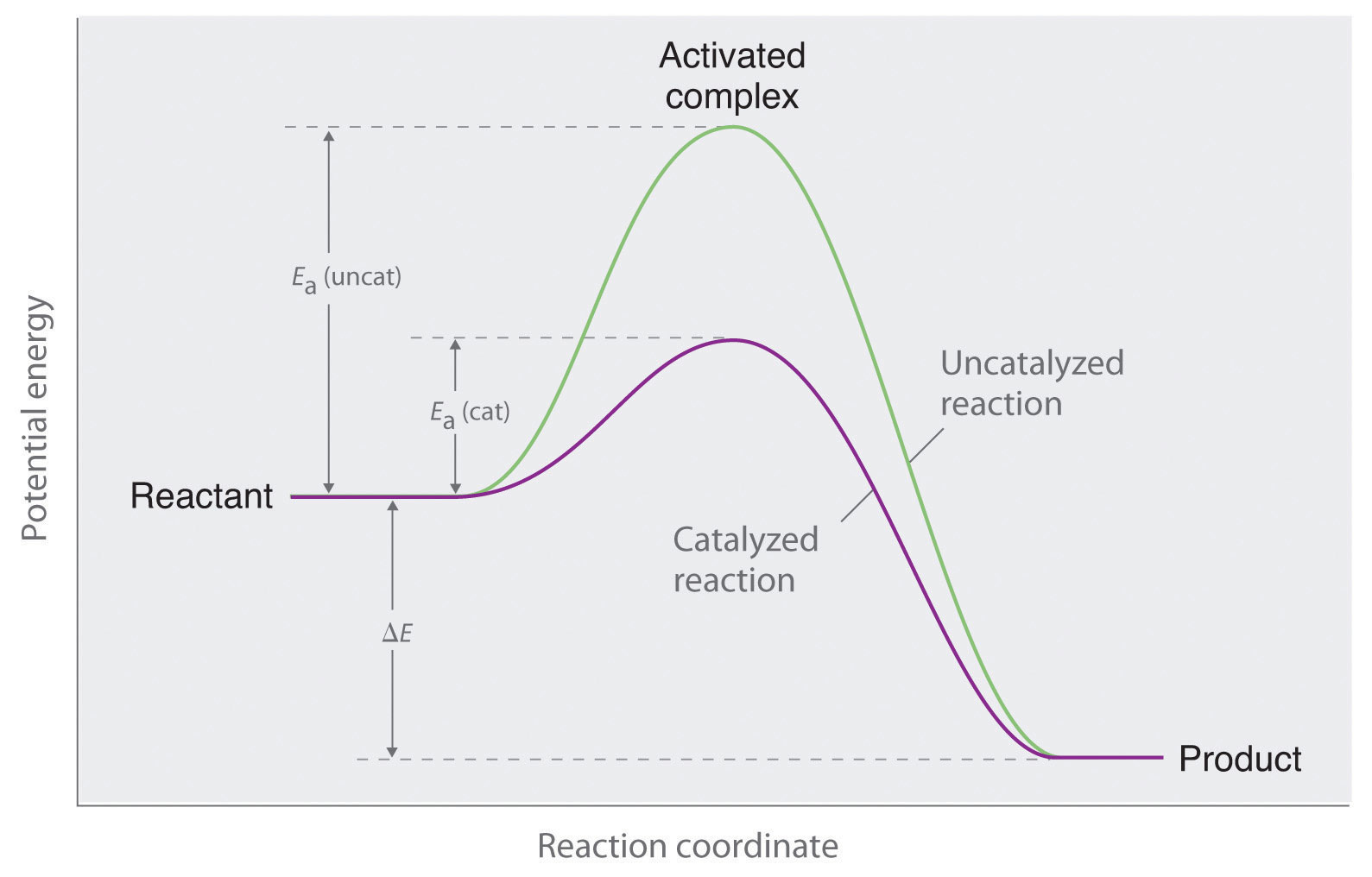
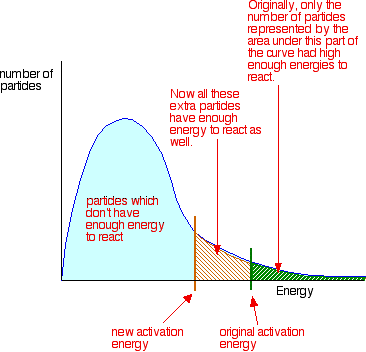
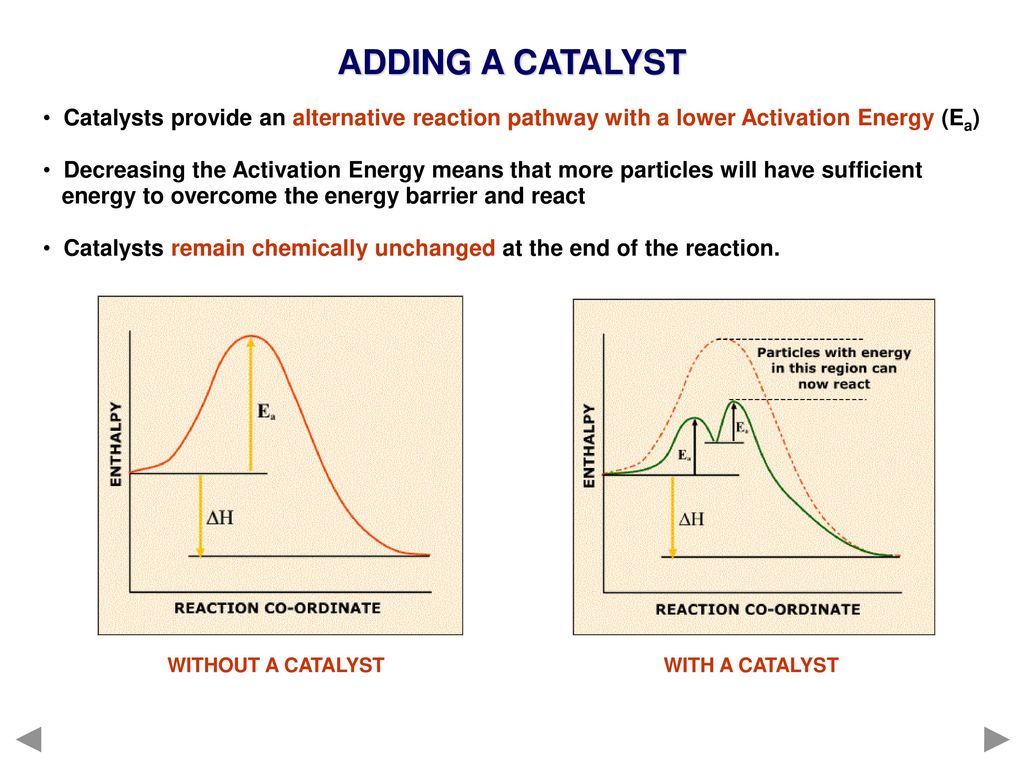
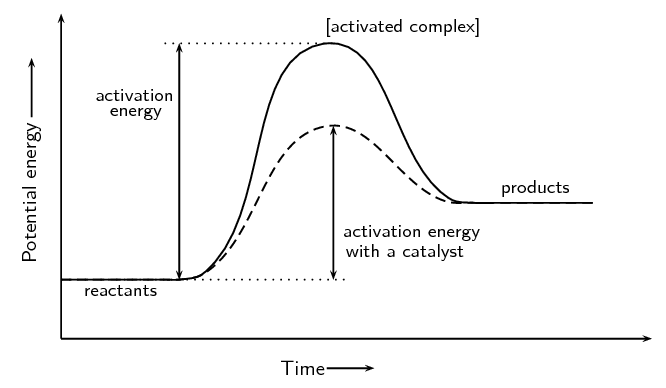
4.19 understand the term 'activation energy' and represent it on a reaction profile 4.21 explain that a catalyst speeds up a reaction by providing an alternative pathway with lower activation energy -

What overall effect does adding a catalyst have on a chemical reaction? It slows the reaction. It speeds - brainly.com

Scientist Adding Catalyst To Boost Chemical Reaction | Presentation Graphics | Presentation PowerPoint Example | Slide Templates

How is the activation energy of a reverse reaction affected by the addition of a catalyst? Is it increased, decreased, or not affected? Explain. | Homework.Study.com

What effect does adding a catalyst have on the rate of a reaction? How does it work on a molecular level? | Homework.Study.com





.png_img_upload_solution_2022-06-01%2007:35:59.682481.png)