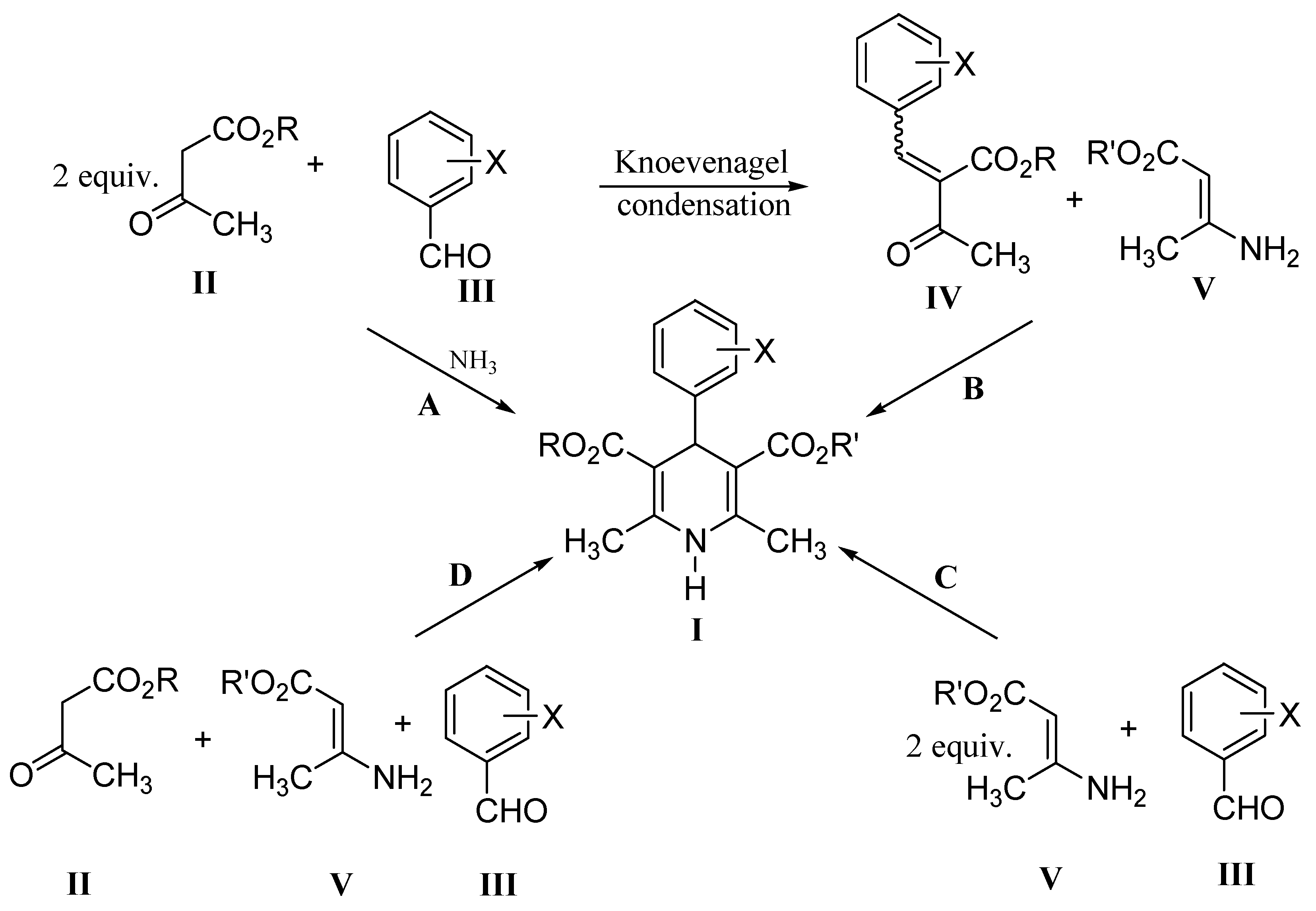
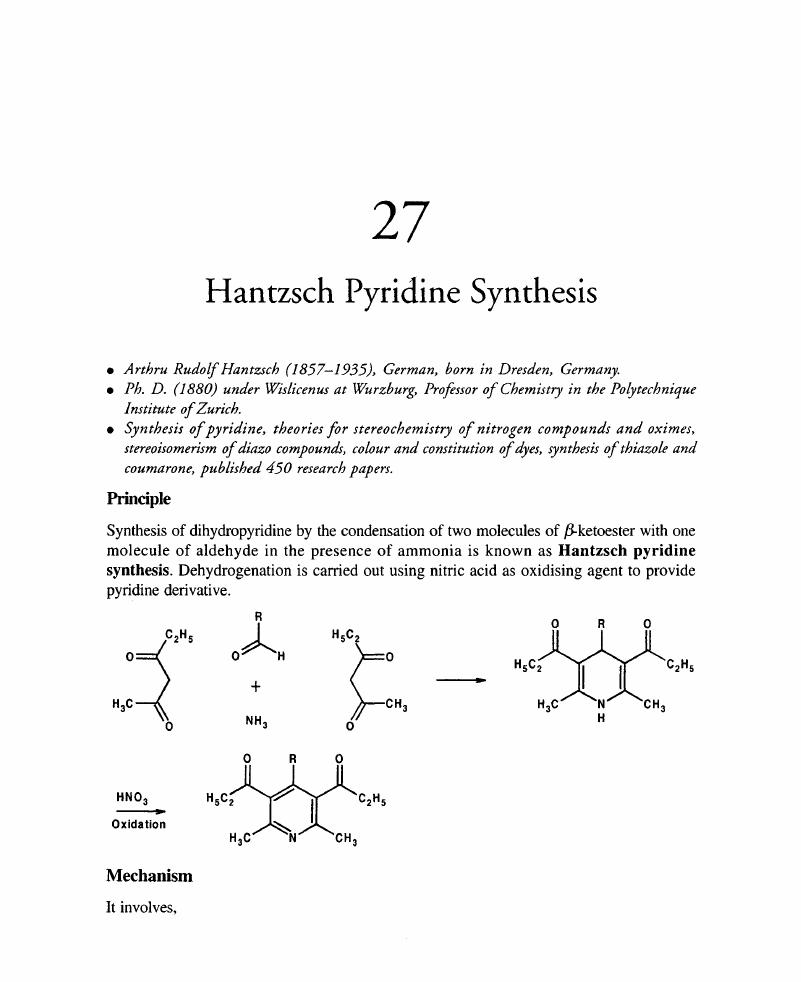
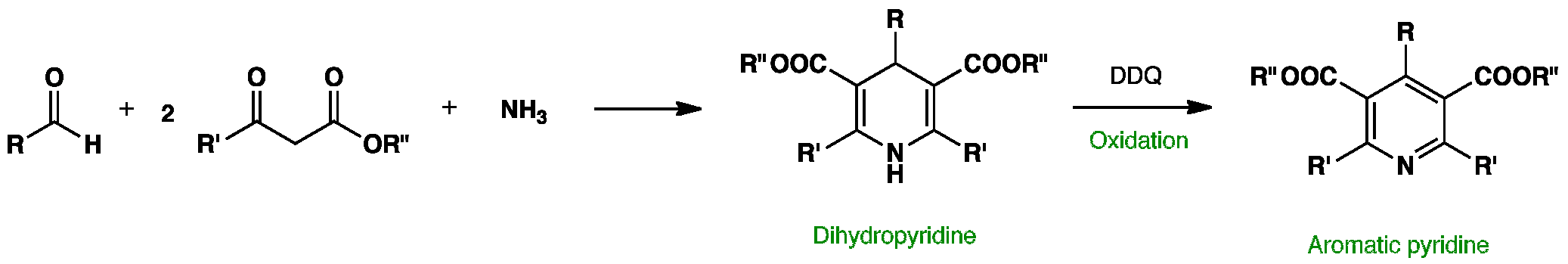
Green approach for synthesis of bioactive Hantzsch 1,4-dihydropyridine derivatives based on thiophene moiety via multicomponent reaction | Royal Society Open Science

Kröhnke-Pyridin-Synthese Hantzsch-Pyridin-Synthese chemische Synthese 2,6-Lutidin, Salz, 26lutidin, Ammoniumacetat, Winkel png | PNGWing