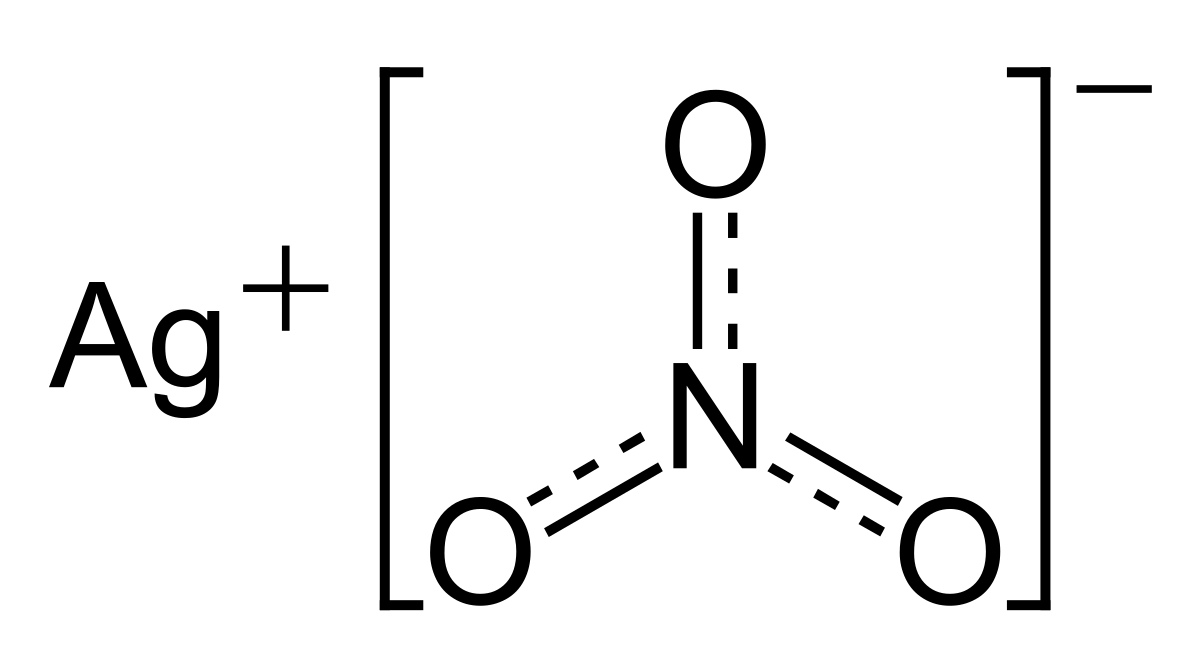
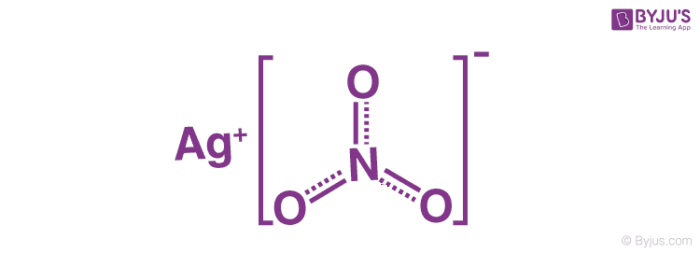
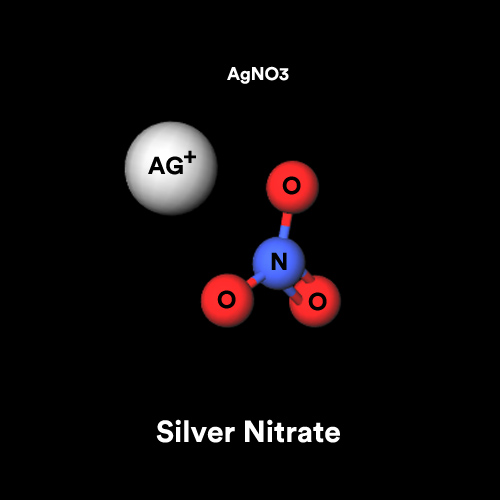
A chemist has 4.0 g of silver nitrate and needs to prepare 2.0 L of a 0.010 M solution. Will there be enough silver nitrtate? If so, how much silver nitrate will

Molar Mass Sections 4-4 to 4-6. Carbon Atomic # is 6 Atomic mass is u Molar mass (mass per mole) is g/mole. - ppt download

The value of observed and calculated molecular weight of silver nitrate are 92.64 and 170 respective - YouTube





![Shows the molecular structure of silver nitrate [16]. | Download Scientific Diagram Shows the molecular structure of silver nitrate [16]. | Download Scientific Diagram](https://www.researchgate.net/publication/350378697/figure/fig1/AS:1023359026466819@1620999213908/Shows-the-molecular-structure-of-silver-nitrate-16.png)