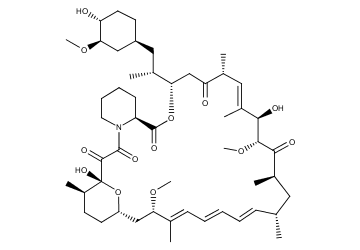
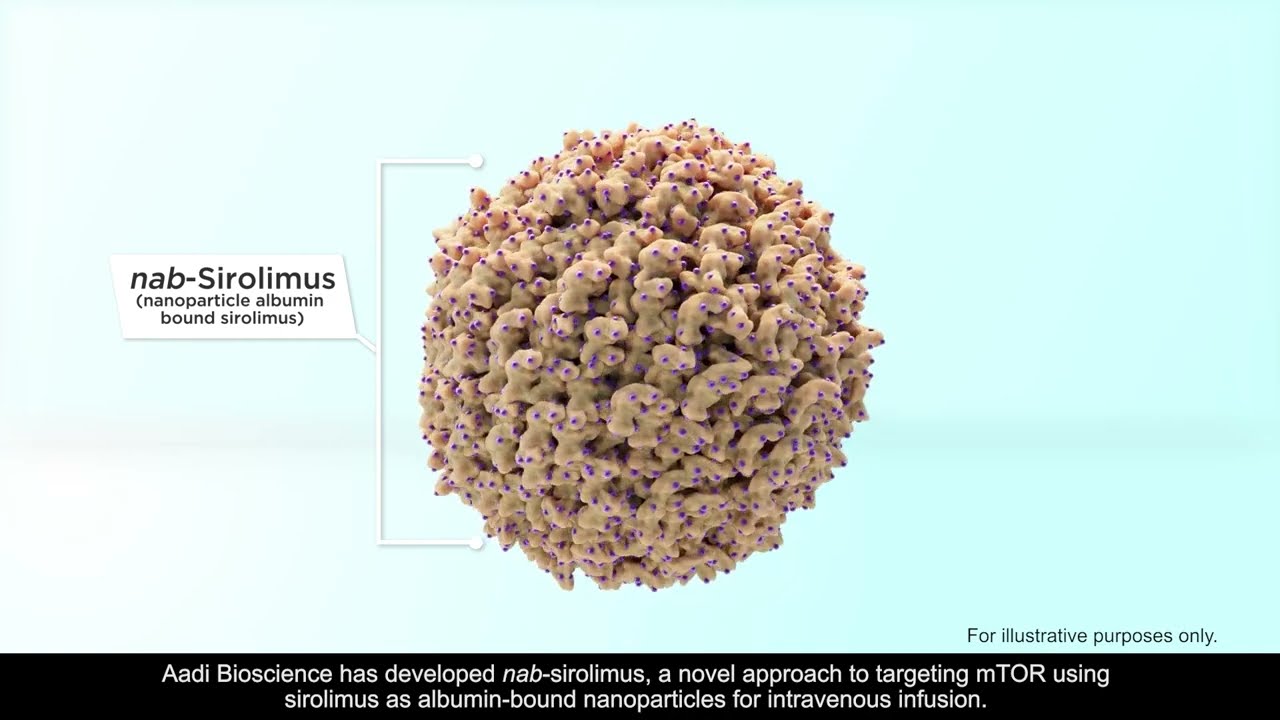
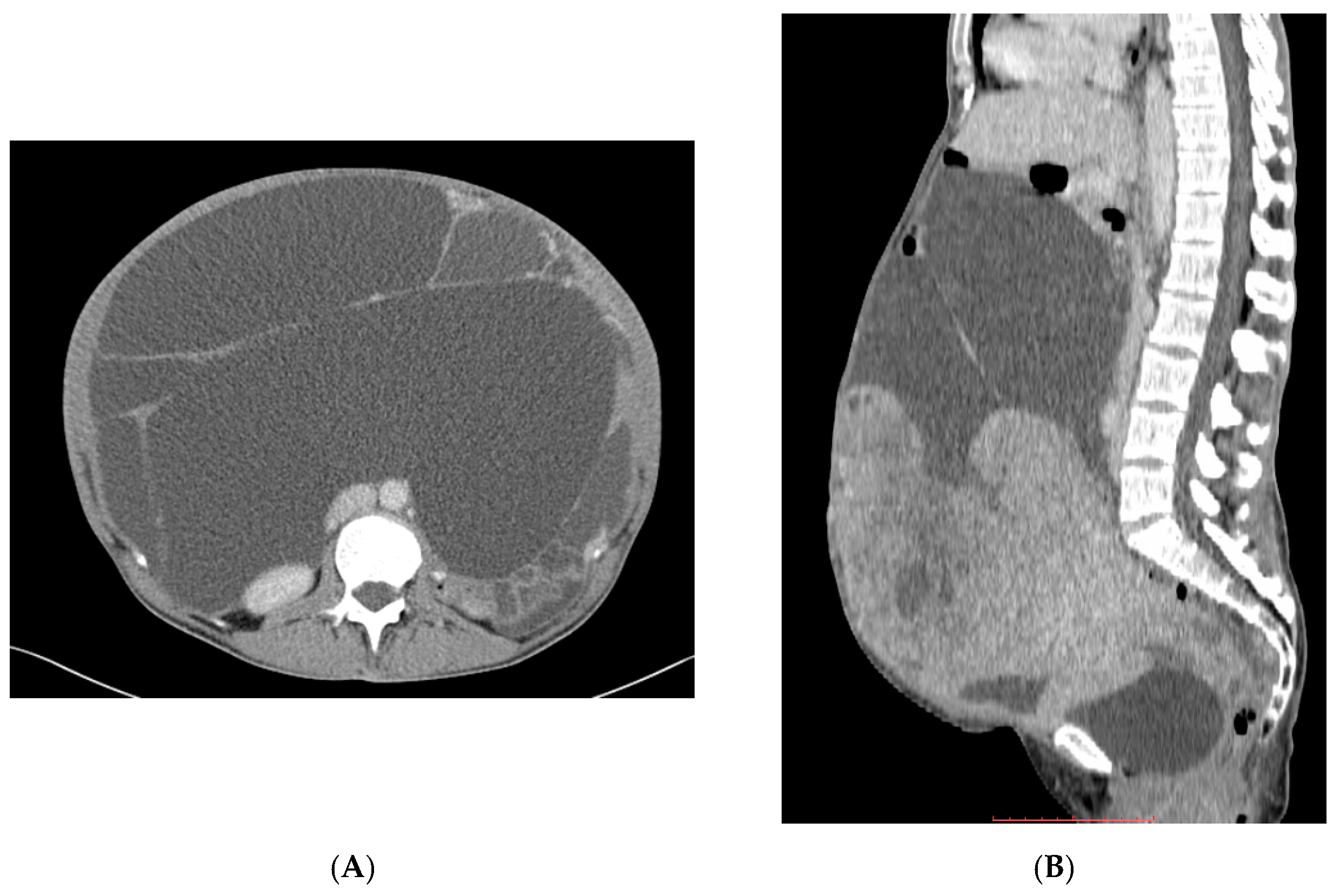
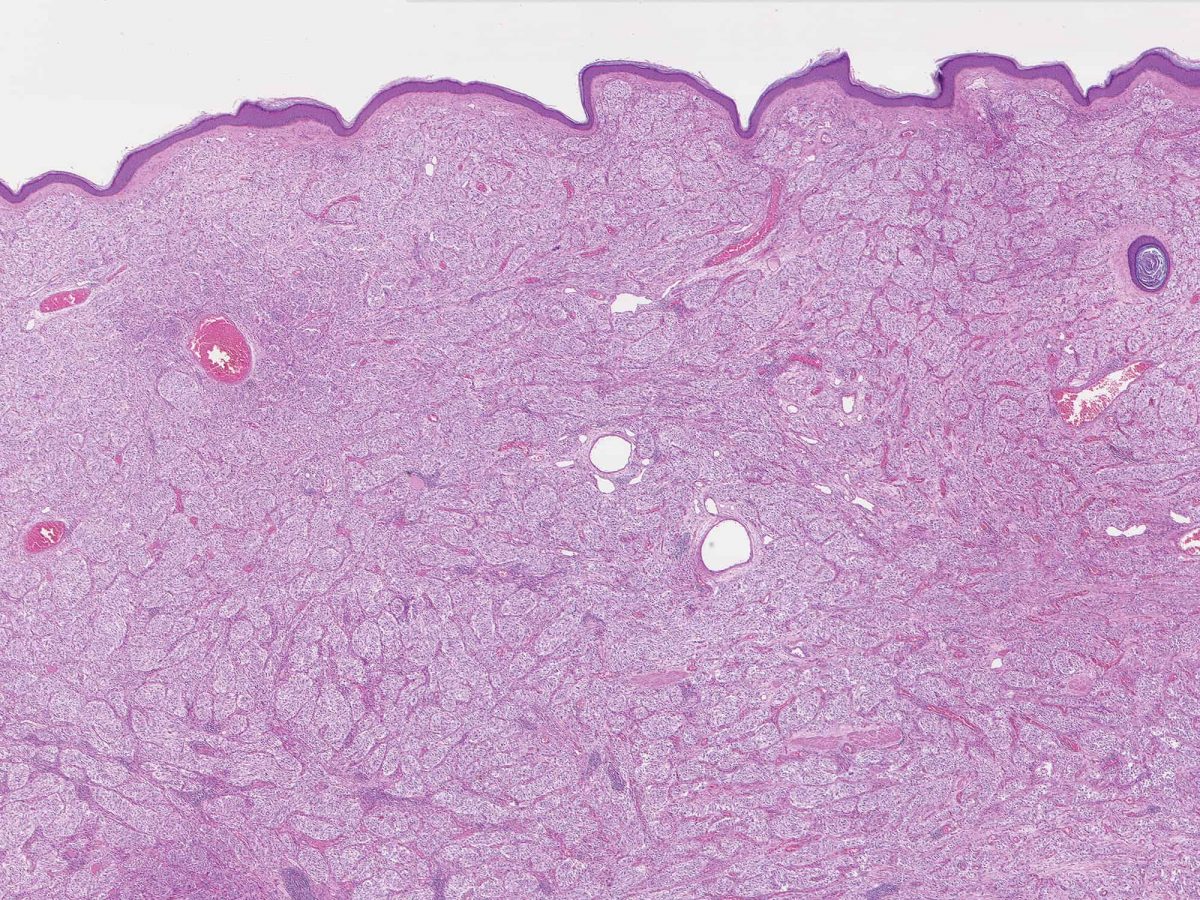
Full article: Nanomedicine approaches for sirolimus delivery: a review of pharmaceutical properties and preclinical studies
ABI-009 (nab-sirolimus) in Advanced Malignant Perivascular Epithelioid Cell Tumors (PEComa): Preliminary Efficacy, Safety, and M

Mechanistic target of rapamycin in the tumor microenvironment and its potential as a therapeutic target for pancreatic cancer - ScienceDirect

Erratum: nab-Sirolimus for Patients With Malignant Perivascular Epithelioid Cell Tumors | Journal of Clinical Oncology
Phase 2, multicenter, open-label basket trial of nab-sirolimus for patients with malignant solid tumors harboring pathogenic ina

Vivek Subbiah, MD on X: "🚨Today FDA approved a drug nab-sirolimus for an ultra rare sarcoma- a unicorn🦄 -PECOMA. 👉🏼Sarcomas are more than 150 different types. 👉🏼Time to identify targets & drugs

Targeted Oncology on X: "FDA approval has been granted to nab-sirolimus for intravenous use as treatment of patients with advanced malignant perivascular epithelioid cell tumors. READ MORE: https://t.co/byWBqVgF38 https://t.co/CN8RVCpeCG" / X
ABI-009 (nab-Sirolimus) Improves Tumor Accumulation and Antitumor Activity over Oral mTOR Inhibitors

Aadi Bioscience Announces Poster Presentations at the North American Neuroendocrine Tumor Society (NANETS) 2023 Multidisciplinary NET Medical Symposium
Fyarro (Sirolimus Protein-gebundene Partikel (Albumin-gebunden)) Online kaufen - Preis & Kosten | Everyone.org
Aadi Bioscience highlights combination of KRAS inhibitors and nab-sirolimus for tumors | Seeking Alpha

nab-Sirolimus for Patients With Malignant Perivascular Epithelioid Cell Tumors | Journal of Clinical Oncology

PDF) Rapid and Durable Response With Nab-Sirolimus After Everolimus Failure in a Patient With Perivascular Epithelioid Cell Tumors (PEComas) of the Uterus

nab-Sirolimus for Patients With Malignant Perivascular Epithelioid Cell Tumors | Journal of Clinical Oncology