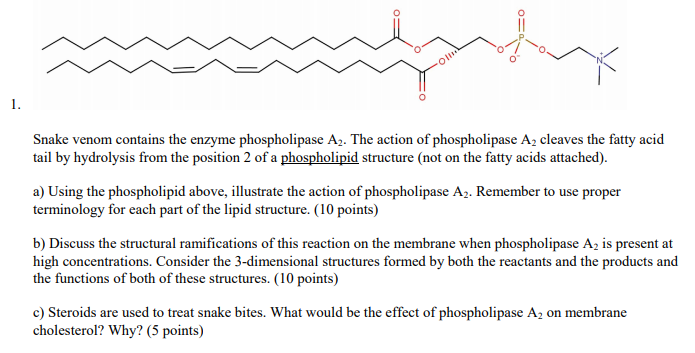
Unraveling snake venom phospholipase A2: an overview of its structure, pharmacology, and inhibitors | Pharmacological Reports
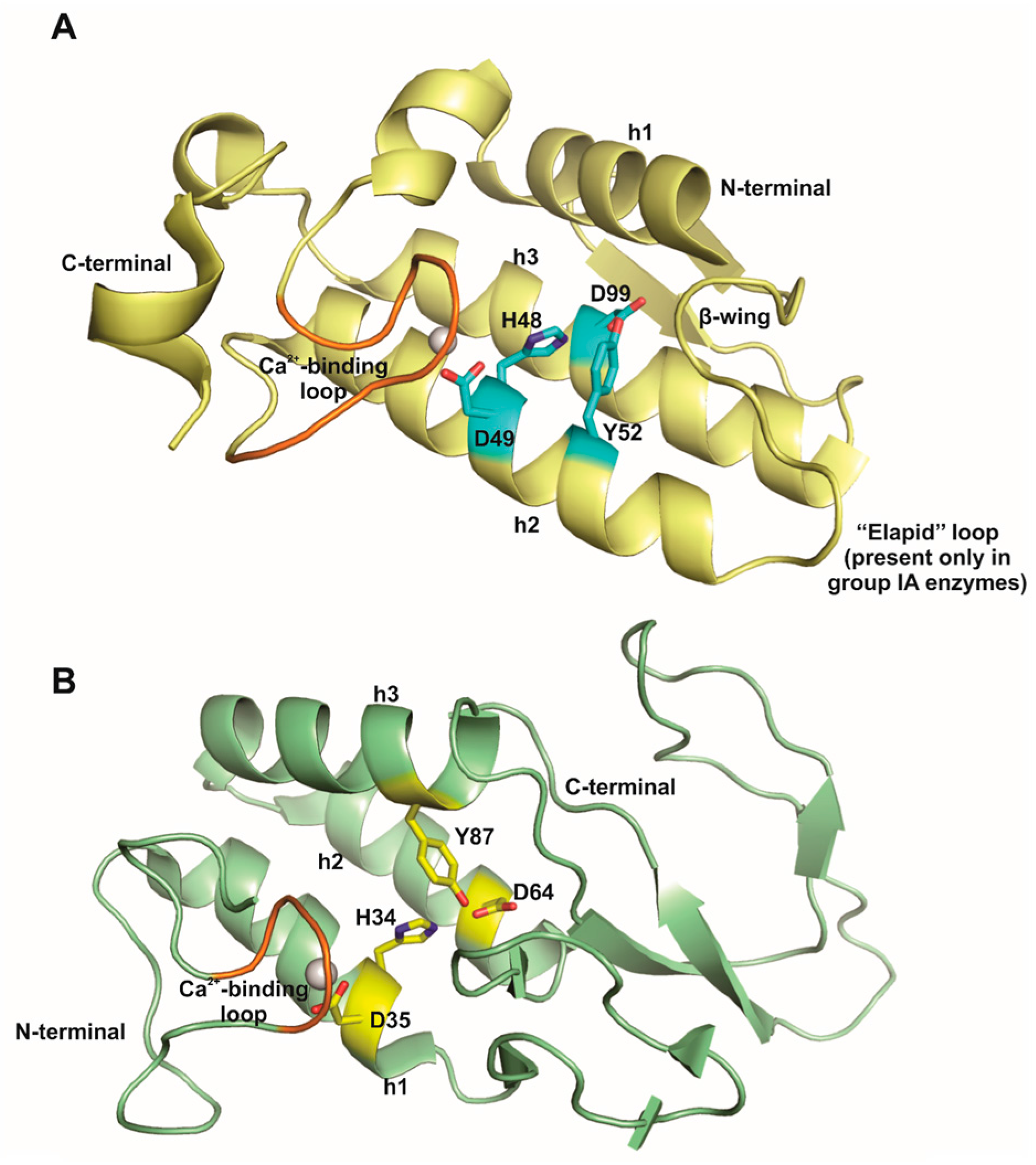
![PDF] Active site of bee venom phospholipase A2: the role of histidine-34, aspartate-64 and tyrosine-87. | Semantic Scholar PDF] Active site of bee venom phospholipase A2: the role of histidine-34, aspartate-64 and tyrosine-87. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7ef426ee07eea4da0943d1d40aa551a47f8ba04d/2-Figure1-1.png)
PDF] Active site of bee venom phospholipase A2: the role of histidine-34, aspartate-64 and tyrosine-87. | Semantic Scholar

Isolation of two basic phospholipases A2 from Bothrops diporus snake venom: Comparative characterization and synergism between Asp49 and Lys49 variants - ScienceDirect

Unraveling snake venom phospholipase A2: an overview of its structure, pharmacology, and inhibitors | Pharmacological Reports

Exploring and understanding the functional role, and biochemical and structural characteristics of an acidic phospholipase A2, AplTx-I, purified from Agkistrodon piscivorus leucostoma snake venom - ScienceDirect
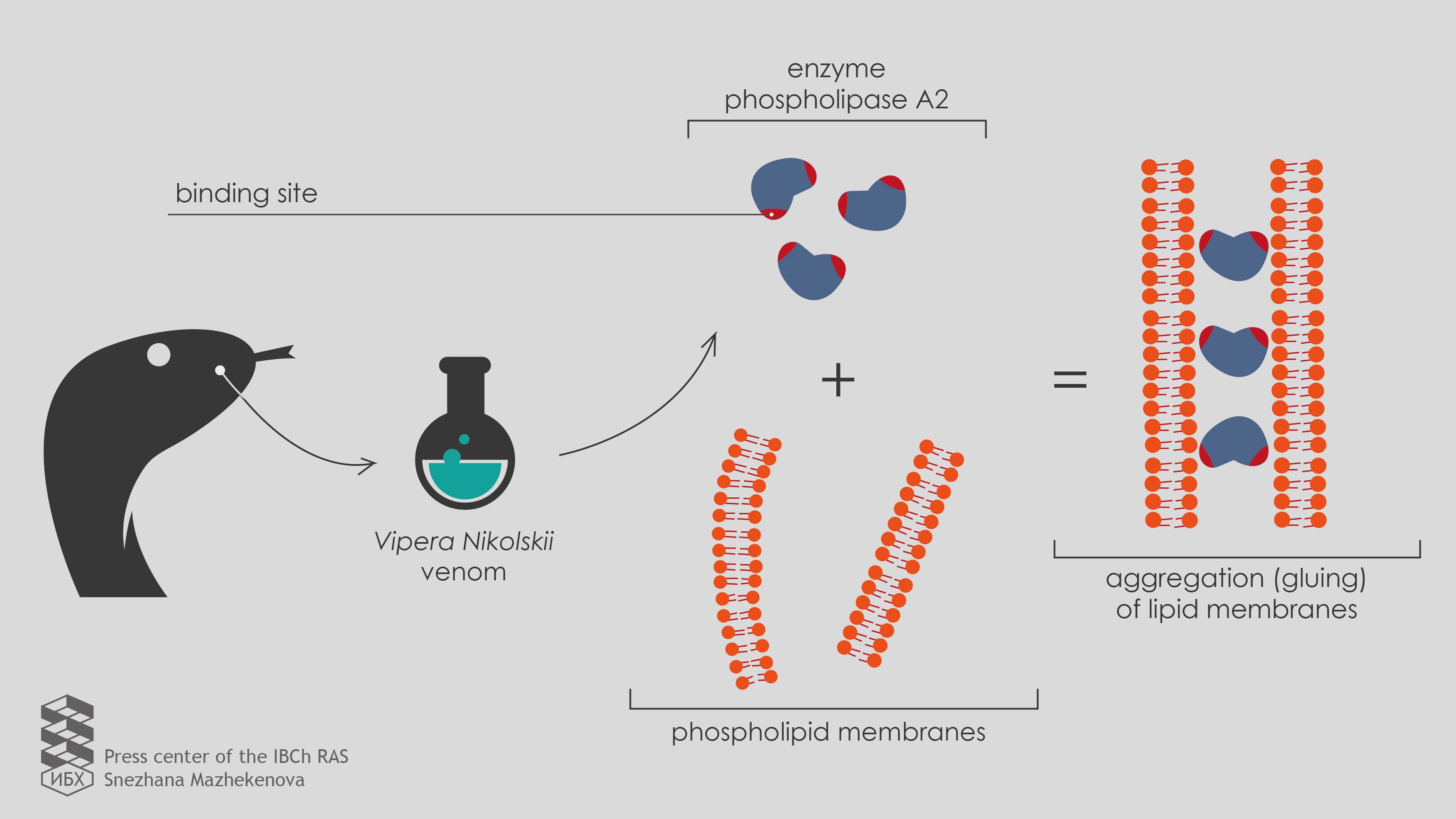
Chemists discovered a new mechanism of action of a Vipera nikolskii venom neurotoxin - Press-room - IBCh RAS
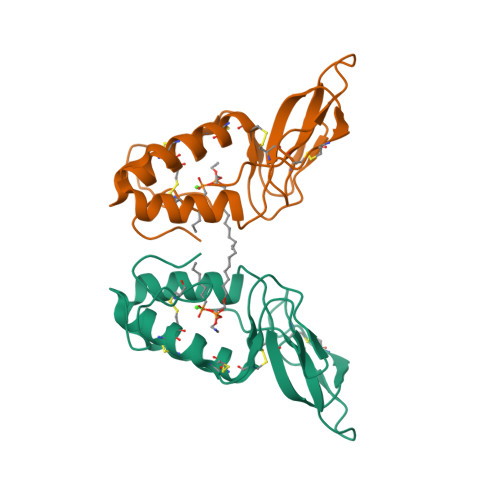
RCSB PDB - 1POC: CRYSTAL STRUCTURE OF BEE-VENOM PHOSPHOLIPASE A2 IN A COMPLEX WITH A TRANSITION-STATE ANALOGUE

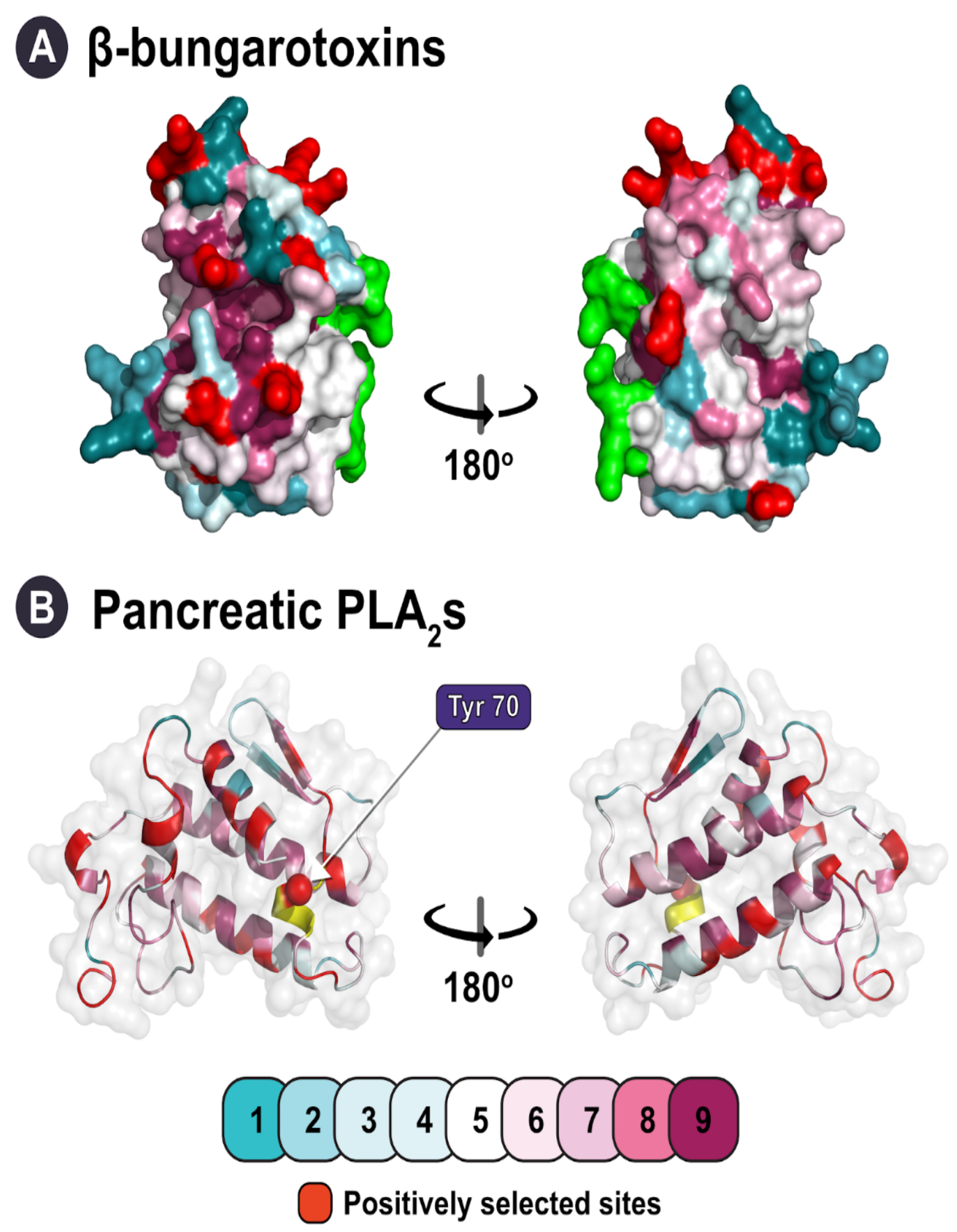
Catalytically Active Snake Venom PLA2 Enzymes: An Overview of Its Elusive Mechanisms of Reaction | Journal of Medicinal Chemistry

Catalytically Active Snake Venom PLA2 Enzymes: An Overview of Its Elusive Mechanisms of Reaction | Journal of Medicinal Chemistry

In Vivo Neutralization of Myotoxin II, a Phospholipase A2 Homologue from Bothrops asper Venom, Using Peptides Discovered via Phage Display Technology | ACS Omega

Panacea within a Pandora's box: the antiparasitic effects of phospholipases A2 (PLA2s) from snake venoms: Trends in Parasitology

Phospholipases A2 from viperidae snake venoms: how do they induce skeletal muscle damage? | Semantic Scholar

Phospholipase A2 enzyme from the venom of Egyptian honey bee Apis mellifera lamarckii with anti-platelet aggregation and anti-coagulation activities | Journal of Genetic Engineering and Biotechnology | Full Text