The standard reduction potential of a silver chloride electrode (metal -sparingly soluble salt - YouTube

Which of the following represents the reduction potential of silver wire dipped into `0.1M AgNO` - YouTube

Electrochemical formation of silver nanoparticles and their applications in the reduction and detection of nitrates at neutral pH | Journal of Applied Electrochemistry

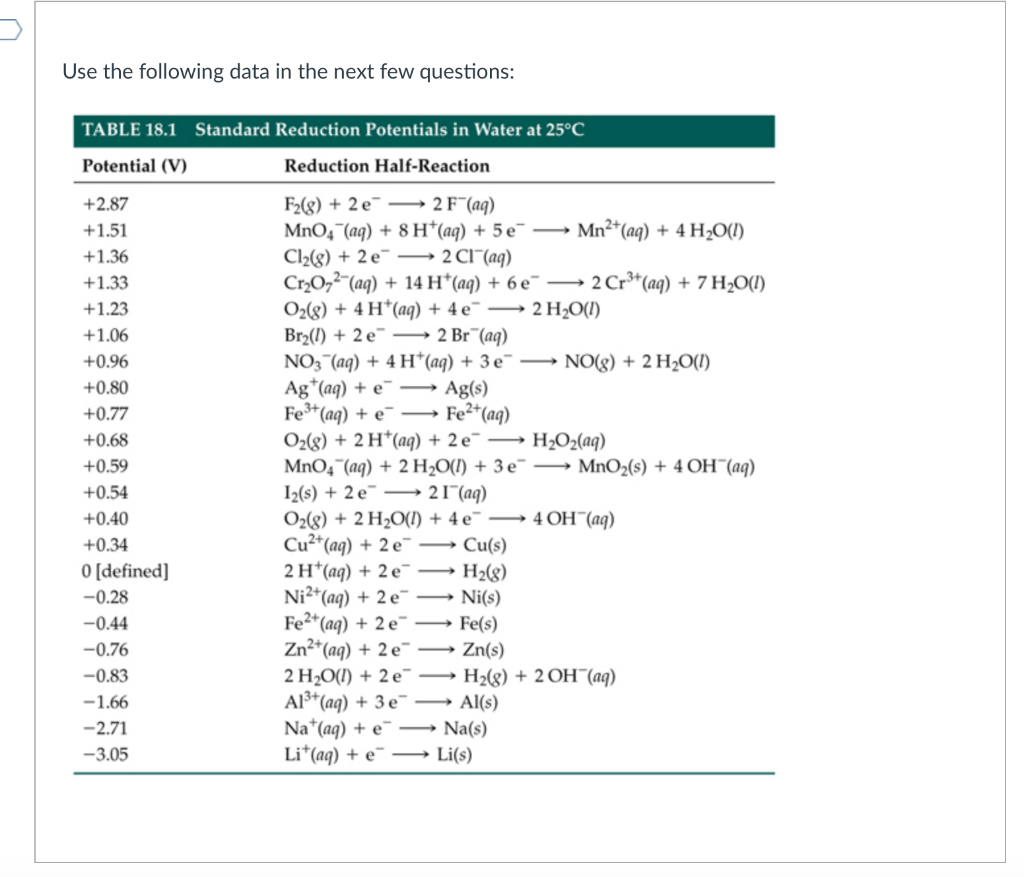
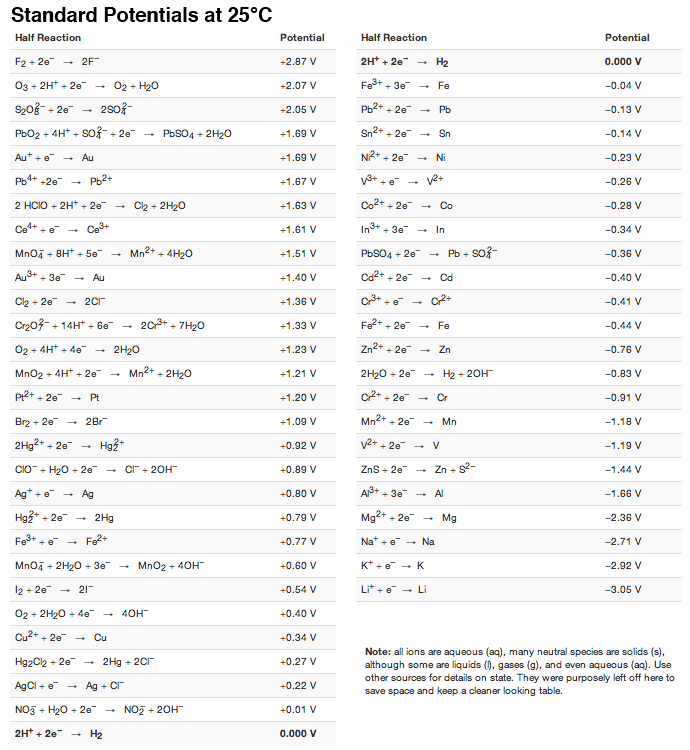
SOLVED: Calculate the standard potential provided by a cell using aluminum, aluminum nitrate, silver; and silver nitrate. Some useful reduction potentials are as follows: Potential Reaction Al3+ + 3e 5 Al Ag +
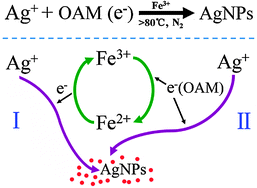
Metal ion redox potential plays an important role in high-yield synthesis of monodisperse silver nanoparticles - Chemical Communications (RSC Publishing)

Standard reduction potential (V vs Ag/AgCl) of different elements in... | Download Scientific Diagram

The standard reduction potential of a Agcl/Ag electrode is 0.2 V and that of a silver electrode (Agt /Ag) is 0.79 V. The maximum amount of AgCl that can dissolve in 10°

Evolution of silver to a better electrocatalyst: Water-assisted oxygen reduction reaction at silver chloride nanowires in alkaline solution - ScienceDirect

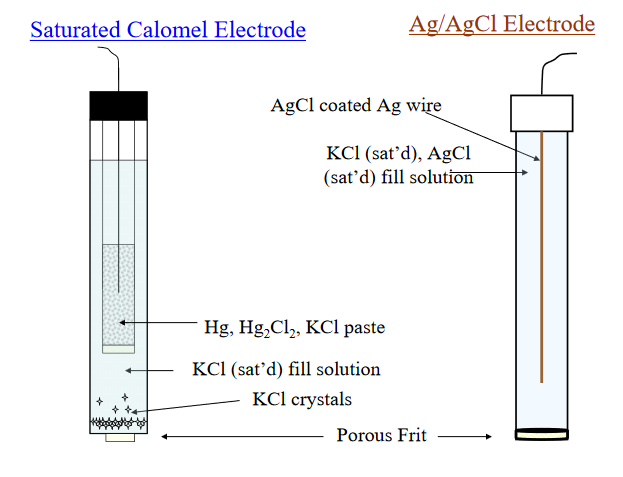
The potential of a silver`//` silver chloride electrode measured with respect to a saturated calomel - YouTube

Question Video: Calculating the Standard Cell Potential for a Magnesium/ Silver Galvanic Cell | Nagwa

Electrocatalytic Nitrate Reduction on Oxide-Derived Silver with Tunable Selectivity to Nitrite and Ammonia | ACS Catalysis
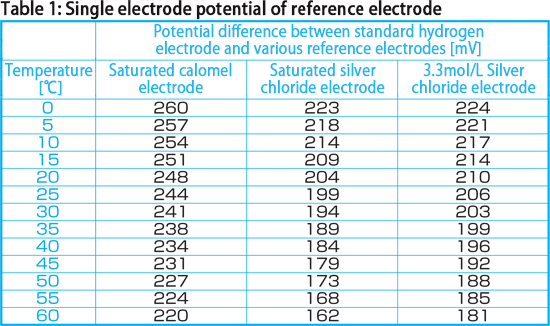
Single electrode potential of reference electrode | Useful information | Product Support | DKK-TOA CORPORATION





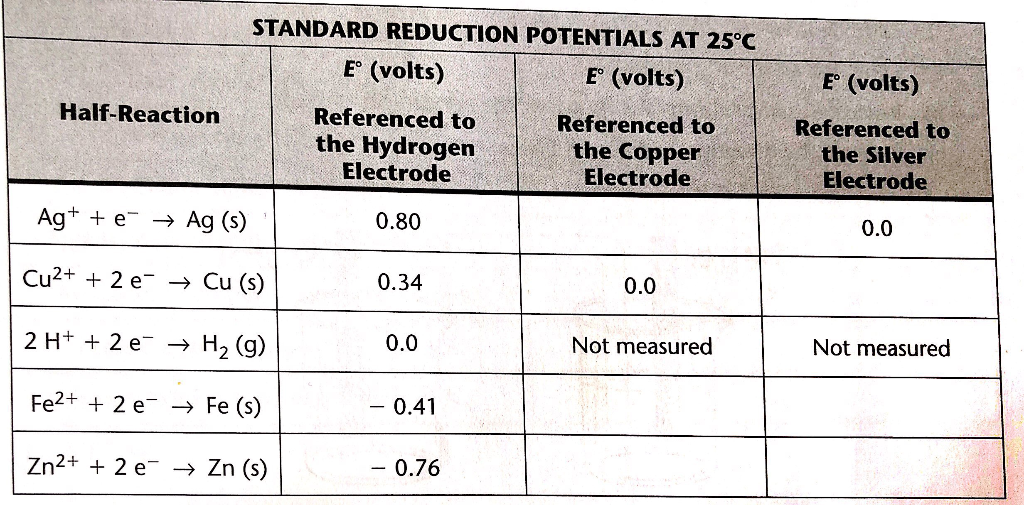
![Standard reduction potentials at 298°K. [24] | Download Table Standard reduction potentials at 298°K. [24] | Download Table](https://www.researchgate.net/publication/316026333/figure/tbl2/AS:650784626708491@1532170554986/Standard-reduction-potentials-at-298K-24.png)